
Back إيساتين Arabic İzatin Azerbaijani Isatin Czech Isatin German Isatina Spanish ایزاتین Persian Isatiini Finnish Isatine French Իզատին Armenian イサチン Japanese
| |

| |
| Names | |
|---|---|
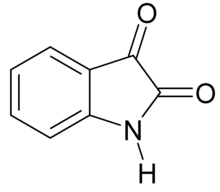
| Preferred IUPAC name
1H-Indole-2,3-dione | |
| Identifiers | |
3D model (JSmol)
|
|
| 383659 | |
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| DrugBank | |
| ECHA InfoCard | 100.001.889 |
| EC Number |
|
| 165206 | |
| KEGG | |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C8H5NO2 | |
| Molar mass | 147.1308 g/mol |
| Appearance | Orange-red solid |
| Melting point | 200 °C (392 °F; 473 K) |
| Hazards | |
| GHS labelling: | |

| |
| Warning | |
| H302, H315, H319, H335 | |
| P261, P264, P270, P271, P280, P301+P312, P302+P352, P304+P340, P305+P351+P338, P312, P321, P330, P332+P313, P337+P313, P362, P403+P233, P405, P501 | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Isatin, also known as tribulin, is an organic compound derived from indole with formula C8H5NO2. The compound was first obtained by Otto Linné Erdman[1] and Auguste Laurent[2] in 1840 as a product from the oxidation of indigo dye by nitric acid and chromic acids.
Isatin is a well-known natural product which can be found in plants of the genus Isatis, in Couroupita guianensis,[3][4] and also in humans, as a metabolic derivative of adrenaline.[5]
It looks like a red-orange powder, and it is usually employed as building block for the synthesis of a wide variety of biologically active compounds including antitumorals,[6] antivirals,[7] anti-HIVs,[8] and antituberculars.[9]
The isatin core is also responsible for the color of “Maya blue” and “Maya yellow” dyes.[10]
- ^ Erdmann, Otto Linné (1840). "Untersuchungen über den Indigo". Journal für Praktische Chemie. 19 (1): 321–362. doi:10.1002/prac.18400190161.
- ^ Laurent, Auguste (1840). "Recherches sur l'indigo". Annales de Chimie et de Physique. 3 (3): 393–434.
- ^ Pinto, A. C. (2001). "The chemistry of isatins: a review from 1975 to 1999". J. Braz. Chem. Soc. 12 (3): 273. doi:10.1590/S0103-50532001000300002.
- ^ Bergman, J. (1988). "The structure and properties of some indolic constituents in Couroupita guianensis aubl". Tetrahedron. 41 (14): 2879. doi:10.1016/S0040-4020(01)96609-8.
- ^ Chiyanzu, I. (2003). "Synthesis and evaluation of isatins and thiosemicarbazone derivatives against cruzain, falcipain-2 and rhodesain". Bioorg. Med. Chem. Lett. 13 (20): 3527–30. doi:10.1016/S0960-894X(03)00756-X. PMID 14505663.
- ^ Mallamo, J.P. (2006). "Structure-guided identification of novel VEGFR-2 kinase inhibitors via solution phase parallel synthesis". Bioorg. Med. Chem. Lett. 16 (8): 2158–62. doi:10.1016/j.bmcl.2006.01.063. PMID 16460933.
- ^ He, Y. (2006). "Design, synthesis, and biological evaluations of novel oxindoles as HIV-1 non-nucleoside reverse transcriptase inhibitors". Bioorg. Med. Chem. Lett. 16 (8): 2109–12. doi:10.1016/j.bmcl.2006.01.066. PMID 16464578.
- ^ Sriram, D. (2005). "Synthesis and evaluation of anti-HIV activity of isatin beta-thiosemicarbazone derivatives". Bioorg. Med. Chem. Lett. 15 (20): 4451–5. doi:10.1016/j.bmcl.2005.07.046. PMID 16115762.
- ^ Bin-Jubair, F.A.S. (2010). "Anti-Tubercular activity of Isatin and Derivatives". Int. J. Res. Pharm. Sci. 1: 113.
- ^ Vuzquez de Agredos-Pascual, M.L. (2011). "From Maya Blue to "Maya Yellow": A Connection between Ancient Nanostructured Materials from the Voltammetry of Microparticles". Angew. Chem. Int. Ed. 50 (25): 5741–4. doi:10.1002/anie.201100921. PMID 21557419.