Back لويسيت Arabic لوئیزیت AZB Lewisit Czech Lewisit German Λεβισίτης Greek Lewisita Spanish لوئیزیت Persian Lewisiitti Finnish Lewisite French Lewisit Hungarian

| |||
| |||
| Names | |||
|---|---|---|---|
| Preferred IUPAC name
[(E)-2-Chloroethen-1-yl]arsonous dichloride | |||
| Other names
Chlorovinylarsine dichloride
2-Chloroethenyldichloroarsine (E)-2-Chlorovinylarsonous dichloride (E)-2-Chlorovinyldichloroarsine Dichloro((E)-2-chlorovinyl)arsine | |||
| Identifiers | |||
3D model (JSmol)
|
|||
| ChemSpider | |||
| MeSH | lewisite | ||
PubChem CID
|
|||
| UNII | |||
| UN number | 2810 | ||
CompTox Dashboard (EPA)
|
|||
| |||
| |||
| Properties | |||
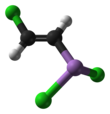
| C2H2AsCl3 | |||
| Molar mass | 207.32 g/mol | ||
| Density | 1.89 g/cm3 | ||
| Melting point | −18 °C (0 °F; 255 K) | ||
| Boiling point | 190 °C (374 °F; 463 K) | ||
| Reacts with water | |||
| Solubility | Ethers, hydrocarbons, THF | ||
| Vapor pressure | 0.58 mmHg (25 °C) | ||
| Hazards | |||
| Occupational safety and health (OHS/OSH): | |||
Main hazards
|
Flammable, highly toxic, corrosive, vesicant | ||
| NFPA 704 (fire diamond) | |||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
Lewisite (L) (A-243) is an organoarsenic compound. It was once manufactured in the U.S., Japan, Germany[2] and the Soviet Union[3] for use as a chemical weapon, acting as a vesicant (blister agent) and lung irritant. Although the substance is colorless and odorless in its pure form, impure samples of lewisite are a yellow, brown, violet-black, green, or amber oily liquid with a distinctive odor that has been described as similar to geraniums.[4][5][6]
Lewisite is named after the US chemist and soldier Winford Lee Lewis (1878–1943). Apart from its use as a weapon of war, the compound is useless; a chemist from the United States Army's chemical warfare laboratories said that "no one has ever found any use for the compound".[7]
- ^ Lewisite I – Compound Summary, PubChem.
- ^ Mitchell, Jon (27 July 2013). "A drop in the ocean: the sea-dumping of chemical weapons in Okinawa" – via Japan Times Online.
- ^ "Russia Completes Destruction of First 10 Tons of Lewisite – Analysis – NTI". www.nti.org.
- ^ Cite error: The named reference
nrcwas invoked but never defined (see the help page). - ^ "CDC – The Emergency Response Safety and Health Database: Blister Agent: Lewsite (L) – NIOSH". www.cdc.gov. Retrieved 2016-01-14.
- ^ Goldman, Max; Dacre, Jack C. (February 14, 1989). Ware, George W. (ed.). "Reviews of Environmental Contamination and Toxicology: Continuation of Residue Reviews". Reviews of Environmental Contamination and Toxicology. 110. Springer: 75–115. doi:10.1007/978-1-4684-7092-5_2. PMID 2692088 – via Springer Link.
- ^ Jarman, Gordon N. (January 1, 1959). "Chemical Corps Experience in the Manufacture of Lewisite". Metal-Organic Compounds. Advances in Chemistry. Vol. 23. American Chemical Society. pp. 328–337. doi:10.1021/ba-1959-0023.ch031. ISBN 978-0-8412-0024-1.