
Back Modelo atomico de Rutherford AN نموذج رذرفورد للذرة Arabic Modelu atómicu de Rutherford AST Ruterford atom modeli Azerbaijani Планетарная мадэль атама Byelorussian Модел на Ръдърфорд Bulgarian রাদারফোর্ড পরমাণু মডেল Bengali/Bangla Model atòmic de Rutherford Catalan Rutherfordův model atomu Czech Rutherfordsches Atommodell German
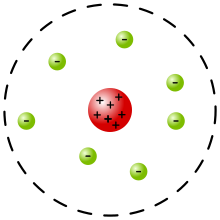
The Rutherford model was devised by Ernest Rutherford to describe an atom. Rutherford directed the Geiger–Marsden experiment in 1909, which suggested, upon Rutherford's 1911 analysis, that J. J. Thomson's plum pudding model of the atom was incorrect. Rutherford's new model[1] for the atom, based on the experimental results, contained new features of a relatively high central charge concentrated into a very small volume in comparison to the rest of the atom and with this central volume containing most of the atom's mass; this region would be known as the atomic nucleus. The Rutherford model was subsequently superseded by the Bohr model.